Figure 1-15 Equilibrium ratio (K) for i-Butane. It is also produced in the process of making gasoline and other fuels from crude oil and making coke from coal.Toluene is used in making paints, paint thinners, fingernail polish, lacquers, adhesives, and rubber and in some printing and leather tanning processes. Toluene occurs naturally in crude oil and in the tolu tree. Figure 1-14 Equilibrium ratio (K) for Propane. Toluene is a clear, colorless liquid with a distinctive smell.

Figure 1-13 Equilibrium ratio (K) for Ethane. K Value charts: Figure 1-12 Equilibrium ratio (K) for Methane. Halvorsen and Sigurd Skogestad, Norwegian University of Science and Technology (scroll down to: 2.2. In the other hand, the DePriester Chart Figure 1-17, may be used for all hydrocarbon components. Equilibrium flash of a multi-component liquid.The present methods are not limited to the. Low temperature: DePriester Chart K values increase wit h decreasing P and increasing T. Concerning heat injection wells, there are various methods for applying heat to the organic-rich rock formation 16. Α = ( y i / x i ) ( y j / x j ) = K i / K j values are widely used in the design of large-scale distillation columns for distilling multi-component mixtures in oil refineries, petrochemical and chemical plants, natural gas processing plants and other industries. Equilibrium ratio or distribution coefficient.
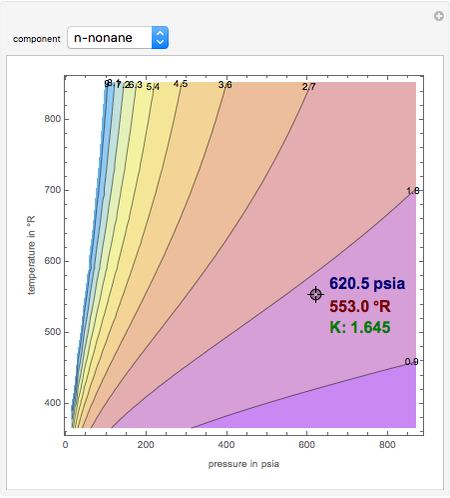
For a liquid mixture of two components (called a binary mixture) at a given temperature and pressure, the relative volatility is defined as If data is not available for the specific gas generalised compressibility charts can be used which require the critical temperature and critical pressure of.
